Abstract
In Drosophila Melanogaster, sweet-responsive gustatory receptor neurons (GRNs) promote feeding, while bitter-responsive GRNs suppress it, but whether appetitive signaling can override aversive signaling remains unclear. This study tested whether optogenetic activation of sweet GRNs could increase proboscis extension response (PER) in the presence of bitter stimulus. Crossed flies expressing csChrimson in sweet GRNs and control flies were subjected to PER assays under three conditions: bitter without light, bitter with red-light activation, and sucrose. Data showed that optogenetic activation increased PER in crossed flies by 269.95%, elevating bitter-evoked PER to levels statistically indistinguishable from sucrose responses. These findings demonstrate that artificial activation of appetitive pathways can override aversion to bitter tastes, and broadly, this study provides insight into how competing sensory modalities are integrated and suggests a potential avenue for research into diabetes and sugar consumption.
Introduction
The gustatory system plays a critical role in feeding behavior by allowing organisms to distinguish between nutritious and toxic substances. In Drosophila melanogaster (fruit flies), taste perception directly drives behavioral outputs such as proboscis extension (feeding) or retraction (avoidance), making it a powerful model for studying how sensory inputs are translated into action (Marella 2006). Gustatory receptor neurons (GRNs) are distributed across multiple peripheral organs, including the proboscis, legs, wings, and internal mouthparts, enabling flies to sample their chemical environment at different spatial locations. These neurons project to the subesophageal ganglion (SOG), the primary taste-processing center in the fly brain, where sensory information is integrated to guide behavior (Wang 2004).
Taste coding in Drosophila is organized into distinct functional pathways that correspond to different taste modalities. GRNs expressing the receptor Gr5a selectively respond to sugars and mediate appetitive behaviors, whereas GRNs expressing Gr66a respond to bitter compounds and drive aversive responses (Marella 2006). This aligns with the labeled-line model of taste coding, in which specific populations of neurons are tuned to particular taste categories and are hardwired to produce defined behavioral outputs (Harris 2015). At the neural level, these pathways remain anatomically segregated: sweet- and bitter-responsive neurons project to distinct regions within the SOG, forming a spatial map in the brain (Wang 2004).
While these pathways have been well characterized in isolation, natural feeding decisions rarely involve exposure to a single taste. Instead, organisms have to integrate competing appetitive and aversive signals to determine whether to consume or reject a food source. We know that the activation of sugar and bitter GRNs via expression of an exogenous ligand-gated ion channel is sufficient to drive taste behaviors, but the extent to which appetitive signaling can counteract or override aversive input remains unclear (Marella 2006). Specifically, it is not known whether artificially enhancing activity in sweet-sensing neurons is enough to bias behavior towards extension in the presence of a bitter stimulus.
Optogenetics provides a powerful approach to address this question by enabling precise, cell-type specific activation of defined neural populations. Channelrhodopsins—light activated ion channels—such as csChrimson allow for selective activation of neurons using light, making it possible to manipulate sensory pathways independent of external chemical stimuli (Klapoetke 2014). csChrimson is a red-shifted excitatory nonselective cation channelrhodopsin that hits peak excitation at 590 nm. It is easily activated by shining a red flashlight onto the flies that express it, as long as light levels are kept low prior to activation. Further, the Chrimson mutation provides white protein to white mutant drosophila, which is important for the bitter sensory neurons to work—the white eye phenotype is expressed in the monoamine transporter for serotonin, which is also released by bitter sensory neurons (Markstein 2018).
This study investigates how sweet and bitter gustatory pathways interact to guide feeding behavior in Drosophila. We tested whether optogenetic activation of sweet GRNs can override the aversive effects of bitter stimuli in a proboscis extension response (PER) assay. We hypothesize that artificial activation of sweet-sensing neurons will increase PER even in the presence of a bitter taste, demonstrating that appetitive signaling can dominate behavioral output when sufficiently enhanced. This would provide insight into how competing sensory modalities are integrated within neural circuits to produce a single behavioral decision.
Methods
Experimental flies were generated by crossing 20xUAS-csChrimson-mVenus females with ;Gr64f-GAL4; Gr64f-GAL4/tm3 males, producing offspring expressing csChrimson in sweet-sensitive GRNs. GR64f was inserted twice on both the second and third chromosome of the males and the second chromosome had no balancer, making it a homozygous viable insertion. The offspring used had the 20xUAS-cs-Chrimson-mVenus/X; Gr64f-GAL4/+; GR64f-GAL4/+ genotype. The GAL4 UAS was used to express the capsaicin sensitive ion channel necessary for the experiment. The w1118 strain of drosophila was used as the control. All crossed flies were fed all-trans retinal (ATR) prior to testing and kept in darkness to prevent premature channel activation.
Flies were anesthetized with CO₂ and mounted onto microscope slides in two rows of five, for ten flies per slide, using minimal nail polish applied to the thorax and wings. While each group glued ten flies down, not all flies survived the process, but PER proportions were calculated as the fraction of responsive flies per condition, so it did not affect data analysis. Following mounting, flies recovered in darkness for 30 minutes to ensure normal neural function. To eliminate thirst as a confound, flies were satiated with water three sequential times. PER responses were then assessed under three conditions: bitter stimulus (50 mM caffeine) without light, bitter stimulus with red light (optogenetic activation), and sweet stimulus (1 M sucrose). Each stimulus was applied to the legs for approximately one second, and PER (extension versus no extension) was recorded. Only flies that responded to sucrose were included in bitter-condition analyses to confirm functional PER capability. The stimuli were all administered in the dark, with two groups using blue light, and the other ten groups using UV light as the baseline before red light was added. This entire protocol was repeated twice: first with the control flies, then with the crossed flies.
Data was analyzed using a two-way ANOVA with strain (control vs cross) and stimulus (bitter no light, bitter light, and sugar) being the factors. Post hoc comparisons were conducted using Bonferroni corrections. We used the data to answer two questions: does optogenetic activation evoke PER in crosses, and does sugar taste activation override bitter?
Results
Proboscis extension response (PER) varied significantly across stimulus conditions and differed between control and experimental flies (Figure 1). A two-way ANOVA revealed a significant effect of stimulus (F(2,66) = 64.116; p < 0.001) and a significant interaction between strain and stimulus (F(2,66) = 16.154; p < 0.001), but a non-significant effect of strain. These results indicate that PER responses depended on the stimulus presented and that the effect of optogenetic activation differed between strains.
Both control and experimental flies showed high PER responses to sucrose, confirming intact feeding behavior across groups (Table 1). Control flies exhibited an average PER of 0.943, while crossed flies had an average PER of 0.763; a nonsignificant difference. Sucrose responses were significantly greater than bitter responses across all groups, confirming that sucrose reliably elicits PER and that all analyzed flies were capable of normal feeding responses. Across both strains, PER was lowest in the bitter no light condition and highest in the sugar condition.
Comparing the bitter conditions without light to bitter with light indicated that optogenetic activation allowed for sugar taste activation to override the bitter taste activation (Figure 2; Table 1). Under bitter conditions without light, control PER was 0.270 ± 0.058 and Crossed PER was 0.213 ± 0.043 and there was no significant difference between the two. For the crossed flies, there was a significant increase of 57.5% in PER between the Bitter Light condition and the Bitter No Light condition (t = 7.513, DF = 66, p < 0.001). In comparison, the control condition’s PER increased a non-significant amount under red-light conditions. Ultimately, under bitter conditions with red-light activation, there was a significant increase of 269.95% from the no-light condition in the crossed PER compared to only a non-significant 42.22% increase in control PER (t = -5.273, DF = 66, p < 0.001). A comparison between the Cross Bitter Light condition and the Cross Sugar conditions reveals that there is a non-significant difference between the two. These results indicate that optogenetic activation of sweet GRNs significantly increases PER in the presence of a bitter stimulus, to the point that it matches the PER of sucrose stimulus.
Data
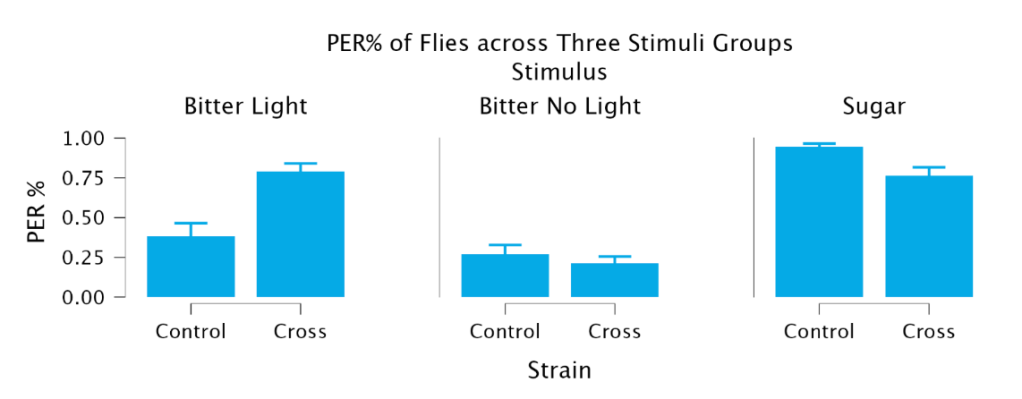
Figure 1. Proboscis extension differed by fly strain only under the Bitter Light condition, where crossed flies showed significantly greater PER than control flies, demonstrating working optogenetic activation.
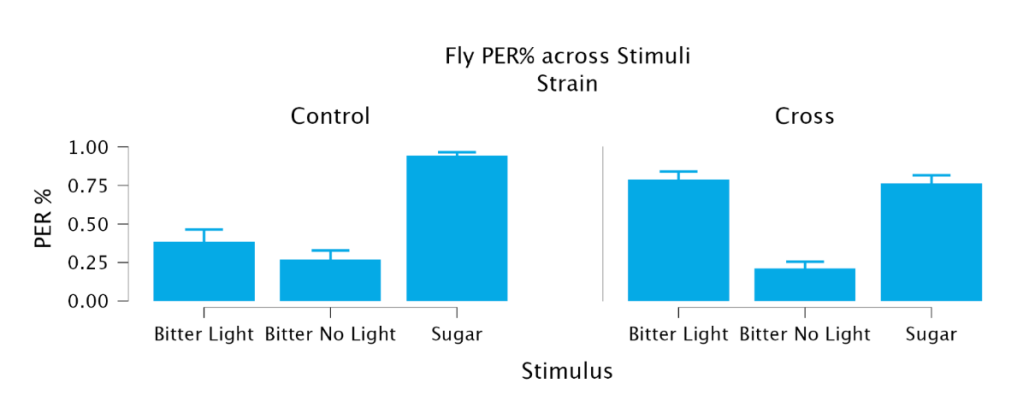
Figure 2. Optogenetic activation of sweet gustatory receptor neurons increased proboscis extension in response to bitter stimuli in crossed flies compared to control flies, indicating that sweet signaling can override bitter-induced aversion.
| Fly Strain | % PER Bitter No Light | % PER Bitter Light | % PER Sugar |
| Control (w1118) | 0.269 ± 0.058 | 0.384 ± 0.080 | 0.943 ± 0.022 |
| Cross | 0.212 ± 0.043 | 0.787 ± 0.052 | 0.763 ± 0.053 |
Table 1. Optogenetic activation increased PER to bitter stimuli in crossed flies while sugar responses were the same in both strains. Data presented as mean ± SEM.
Discussion
We investigated how competing sweet and bitter gustatory pathways are integrated to guide feeding behavior in drosophila and found that optogenetic activation of sweet GRNs significantly increased PER in response to a bitter stimulus, demonstrating that appetitive signaling can override aversive input when artificially enhanced.
The experiment had several limitations. Human error played its part: variability in water satiation between groups could have altered the PER measurements, the use of blue light instead of UV light in two of the twelve trials may have unintentionally activated optogenetic channels, and the amount of time red light was shone on the flies could have activated some sugar GRNs more than others. Genetically, the lack of a matched control lacking csChrimson limited the ability to isolate the effect of optogenetic activation from background genetic differences—the w1118 control flies differed quite a bit from the crossed flies. Finally, the experiment was not conducted blind, introducing possible observer bias from people who knew what the hypothesis and experiment was. Future studies should incorporate double-blind procedures, since PER scoring is subjective.
While baseline PER to bitter stimuli was low in both the control and crossed groups, activation of sweet GRNs nearly quadrupled PER to bitter stimuli in crossed flies. While sucrose responses were significantly greater than bitter responses across all groups, confirming that it reliably elicited PER, the PER responses to sucrose were non-significantly lower in the crossed flies than in the control flies, reflecting possible altered sensitivity due to chronic csChrimson expression in the crossed flies. The optogenetic activation ultimately increased the crossed flies’ PER response to bitter to about equal levels to their response to sugar, indicating that our hypothesis was correct.
These findings align with previous literature showing that the activation of specific GRN populations is sufficient to drive behavior, and extends on those findings by demonstrating that appetitive pathways can actively suppress aversive responses rather than simply operate in parallel (Marella 2006). An interesting implication of these findings is in their relevance to diabetes research. In drosophila, high-sugar diets can help model aspects of Type II diabetes, and the finding that appetitive signaling can override taste responses to bitter raises the possibility that taste circuits could be manipulated to promote consumption of healthier but normally avoided food (Musselman 2011). If flies can be altered to perceive bitter foods as rewarding via activation of sugar GRNs, this could theoretically allow flies to consume healthier foods while still receiving neural rewards associated with sugar. Reduction of sugar in the diet is healthy, as chronic high-sugar diets can reshape sweet taste sensitivity and promote obesity (May 2019). Future research should look further into this pathway, as insulin signaling is highly conserved between flies and humans and this research provides a conceptual model for studying diabetes in humans.
Bibliography
Harris, David T., Kallman, Benjamin R., Mullaney, Brendan C., & Scott, K. (2015). Representations of Taste Modality in the Drosophila Brain. Neuron, 86(6), 1449–1460. https://doi.org/10.1016/j.neuron.2015.05.026.
Klapoetke, N. C., Murata, Y., Kim, S. S., Pulver, S. R., Birdsey-Benson, A., Cho, Y. K., Morimoto, T. K., Chuong, A. S., Carpenter, E. J., Tian, Z., Wang, J., Xie, Y., Yan, Z., Zhang, Y., Chow, B. Y., Surek, B., Melkonian, M., Jayaraman, V., Constantine-Paton, M., & Wong, G. K.-S. (2014). Independent optical excitation of distinct neural populations. Nature Methods, 11(3), 338–346. https://doi.org/10.1038/nmeth.2836.
Marella, S., Fischler, W., Kong, P., Asgarian, S., Rueckert, E., & Scott, K. (2006). Imaging Taste Responses in the Fly Brain Reveals a Functional Map of Taste Category and Behavior. Neuron, 49(2), 285–295. https://doi.org/10.1016/j.neuron.2005.11.037.
Markstein, M. (2018). Drosophila Workers Unite! A Laboratory Manual for Working with Drosophila.
May, C. E., Vaziri, A., Lin, Y. Q., Grushko, O., Khabiri, M., Wang, Q.-P., Holme, K. J., Pletcher, S. D., Freddolino, P. L., Neely, G. G., & Dus, M. (2019). High Dietary Sugar Reshapes Sweet Taste to Promote Feeding Behavior in Drosophila melanogaster. Cell Reports, 27(6), 1675-1685.e7. https://doi.org/10.1016/j.celrep.2019.04.027.
Wang, Z., Singhvi, A., Kong, P., & Scott, K. (2004). Taste Representations in the Drosophila Brain. Cell, 117(7), 981–991. https://doi.org/10.1016/j.cell.2004.06.011.


Leave a comment